The first quarter of 2026 brings a focused leap toward improving quality control, workflow efficiency, and compliance readiness. These LIS updates are designed to eliminate operational gaps, reduce manual dependencies, and ensure laboratories deliver faster, more accurate, and reliable results, without added complexity.
Let’s explore what’s new and how each enhancement drives real impact.
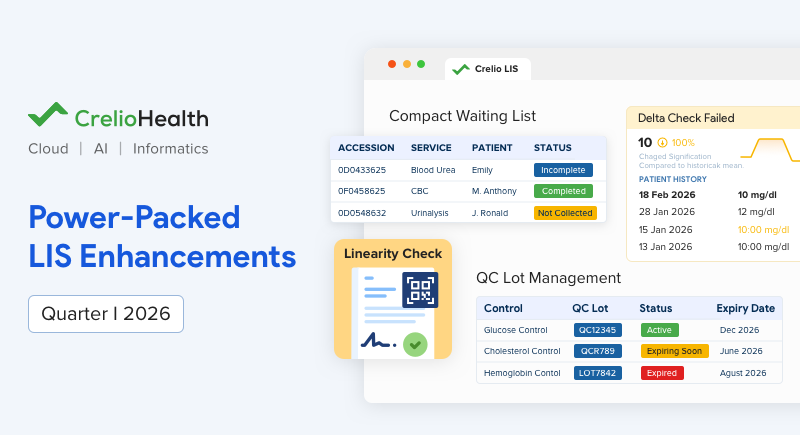
1. Fasten & Simplify Operations With Compact Waiting List
The Compact Waiting List introduces a more structured and condensed view of pending samples and patient workflows, enabling labs to manage waiting lists more efficiently within limited screen space.
Importance of the Feature
Traditional waiting lists can become overwhelming, especially in busy labs handling hundreds of samples daily. A cluttered interface slows down decision-making and increases the chances of missed or delayed actions. This feature ensures that critical tasks remain visible and manageable at all times. A compact and organized view helps teams act faster with better clarity.
Key Takeaways & Benefits
- Clear and concise view of pending workflows
- Faster identification and action on waiting samples
- Reduced navigation and screen clutter
- Improved turnaround time (TAT)
- Better operational focus for lab staff
Use Case: During peak hours, a lab technician managing a long waiting list can switch to Compact View to see more patients and pending reports on a single screen, helping them prioritize work faster and avoid missing items.
2. Manage QC Lot Seamlessly With a Dedicated Module
The QC Lot Management module provides a dedicated space to create, organize, and track quality control lots in a structured and centralized manner within the LIS.
Importance of the Feature
Maintaining QC lot data manually or across scattered systems can cause inconsistencies, incorrect lot usage leading to compliance risks and audit challenges. A dedicated module ensures proper management to ensure that quality control processes remain accurate, traceable, and audit-ready.
Key Takeaways & Benefits
- Centralized and structured QC lot management
- Improved traceability of QC data
- Reduced risk of incorrect or outdated lot usage
- Reduced manual errors in QC data handling
- Improved consistency in quality assurance processes
3. Maintain Result Precision With Linearity Check
The Linearity Check feature ensures that test results remain accurate across a mandated measurable range, supporting reliable diagnostics and compliance with standards.
Importance of the Feature
Ensuring linearity is essential for maintaining the reliability of test results. Without proper linearity validation, test results may drift outside acceptable ranges, leading to inaccurate reporting and potential clinical risks. Any deviation can compromise the accuracy of the results and impact compliance with regulatory standards.
Key Takeaways & Benefits
- Ensures high accuracy and consistency of test results
- Helps detect inconsistencies early
- Reduces the risk of incorrect diagnostics
- Strengthens compliance with regulatory standards
- Builds trust in lab reporting quality
4. Strengthen Patient Safety With Delta Check Validation
The enhanced Delta Check adds an additional validation layer by comparing current test results with previous patient records to identify significant deviations.
Importance of the Feature
Unexpected variations in patient results can indicate errors, sample mix-ups, or genuine clinical changes. Without validation, such anomalies may go unnoticed. Delta checks ensure every significant deviation is reviewed before reporting.
Key Takeaways & Benefits
- Early identification of abnormal result variations
- Automated comparison with historical patient data
- Reduced the risk of reporting inaccurate results
- Improved patient safety and diagnostic accuracy
5. Automate Reporting Workflows With Integrated Cell Counter
The Integrated Cell Counter feature streamlines the process of capturing and recording cell count data directly within the LIS, reducing manual intervention in reporting workflows.
Importance of the Feature
Cell counting processes often involve repetitive manual data entry, which increases the chances of errors and slows down reporting. By integrating this workflow into the LIS, labs can ensure faster, more reliable data handling while eliminating transcription mistakes.
Key Takeaways & Benefits
- Eliminates manual intervention in cell count data handling
- Speeds up reporting workflows and turnaround time
- Improved accuracy by eliminating transcription errors
- Seamless analyzer-to-LIS integration
- Final Thoughts
These 2026 Quarter I updates are not just feature additions; they are workflow enablers. These updates focus on what matters most to modern laboratories: accuracy, efficiency, and compliance. From better workflow visibility and structured QC management to enhanced validation mechanisms and reporting automation, these updates collectively help labs operate with greater confidence and control. Each feature is designed not just to improve processes, but to enable laboratories to deliver faster, safer, and more reliable diagnostic outcomes.