Why Sample Barcode Tracking Is a Clinical Lab’s Best Quality Investment
Picture this: A patient comes in fasting since the previous night for a lipid panel. The phlebotomist is running behind. Labels are printed in a...
Why Enterprise Labs Are Shifting From Multi-Vendor Stacks To Integrated SaaS LIMS
Enterprise labs running 5, 10, or even 50+ collection centers are dealing with a very different reality than smaller labs. Managing multiple vendors across locations,...
From QC Gaps to Inspection-Ready: A Lab Quality Control Blueprint
Quality control in a clinical lab is not simply about running controls and recording results. A mature lab quality control strategy is systematic, documented, and...
10 Critical Questions For LIS Vendor Evaluation Before Signing
The LIS you choose today will directly impact your lab’s efficiency, compliance, and scalability for years to come. Yet, many LIS vendor evaluation processes fall...
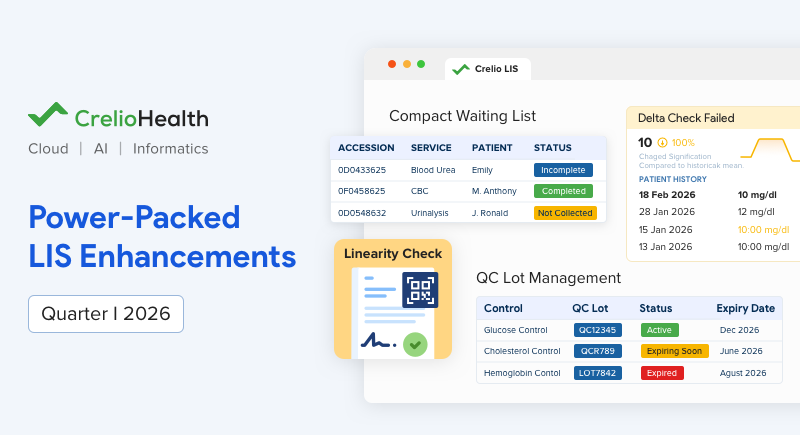
Smarter, Faster, Better: Transformative LIS Updates – Q1 2026
The first quarter of 2026 brings a focused leap toward improving quality control, workflow efficiency, and compliance readiness. These LIS updates are designed to...
From Manual Operations To Smart Integrated Workflows: The Transformation Of Arpan Diagnostic Centre
Dr. Anil Savaliya, Owner and pathologist at Arpan Diagnostics Centre, Rajkot, shares how CrelioHealth lab interfaces make an extraordinary difference in their daily...
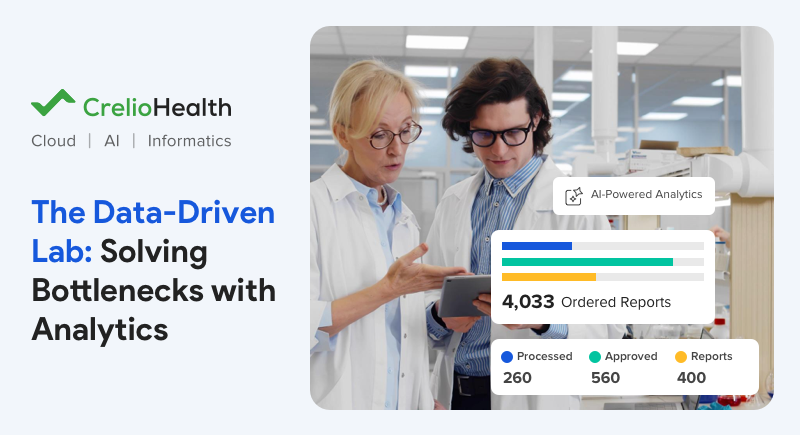
Leveraging Intelligent Lab Data Analytics for Bottleneck-Free Operations
Modern laboratory workflows have become increasingly complex, driven by rising test volumes, multi-instrument environments, and distributed operations. Parallely,...
Why Generic LIMS Fail Veterinary Pathology Labs: A Guide to Specialized Pathology Workflows
Generic LIMS are designed for human healthcare, yet many veterinary pathology labs still rely on them. However, veterinary diagnostics is increasingly complex in...